 
the standard hydrogen electrode) for battery operation. 5 Third, considering the rather narrow operating window beyond which gas (H 2 and O 2) evolution occurs in water, 6 Zn has a suitable redox potential of −0.76 V ( vs. 3 Second, under mildly acidic conditions (pH = 4–6), Zn is oxidized to Zn 2+ without forming intermediate phases 4 and exhibits a high overpotential for the hydrogen evolution reaction (HER). First, not only is it readily accessible and relatively cheap as a resource, but it is also known for its non-toxicity and chemical stability in aqueous media compared to alkali metals such as lithium or sodium. Zn offers several advantages in aqueous electrochemical systems. Aside from the aqueous electrolyte, a distinguishing aspect of this system is the use of Zn metal as the anode. A typical AZIB consists of the following components: Zn metal (anode), an aqueous electrolyte (mildly acidic pH), a separator, and an (in)organic cathode material. 2 Hence, aqueous batteries are much more likely to be able to sustain high rate battery operation.Īmong numerous candidates for aqueous batteries, aqueous zinc (Zn) ion batteries (AZIBs) have recently emerged into the spotlight as promising systems that can meet today's demanding performance levels. Second, water has a significantly higher ionic conductivity than most organic solvents, usually by 2–3 orders of magnitude. Aqueous electrolytes can circumvent this issue. The organic electrolyte is known to act as fuel for such fires, 1 exacerbating their consequences. In the unfortunate scenario where physical/electrical/chemical factors subject a battery to short circuit, the sudden exothermic reactions may result in a fire. First, replacing conventional organic electrolytes with aqueous ones mitigates the risk of fire hazards. Therefore, batteries that target ESS applications must meet two crucial requirements: high power density and safety.Īqueous batteries are extremely promising in this regard. Furthermore, as ESSs consist of multiple battery modules, what may start as a small fire may not only incur heavy costs but also end up having devastating consequences on their surroundings and/or human lives. 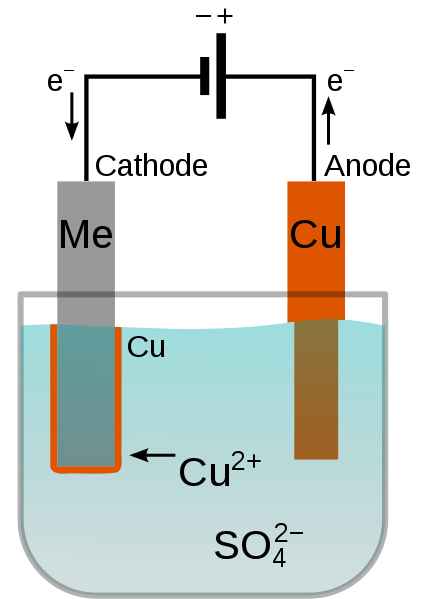
In other words, high rate capability is crucial for such responsiveness, where ESSs can store extra power and discharge when needed in a short period of time. 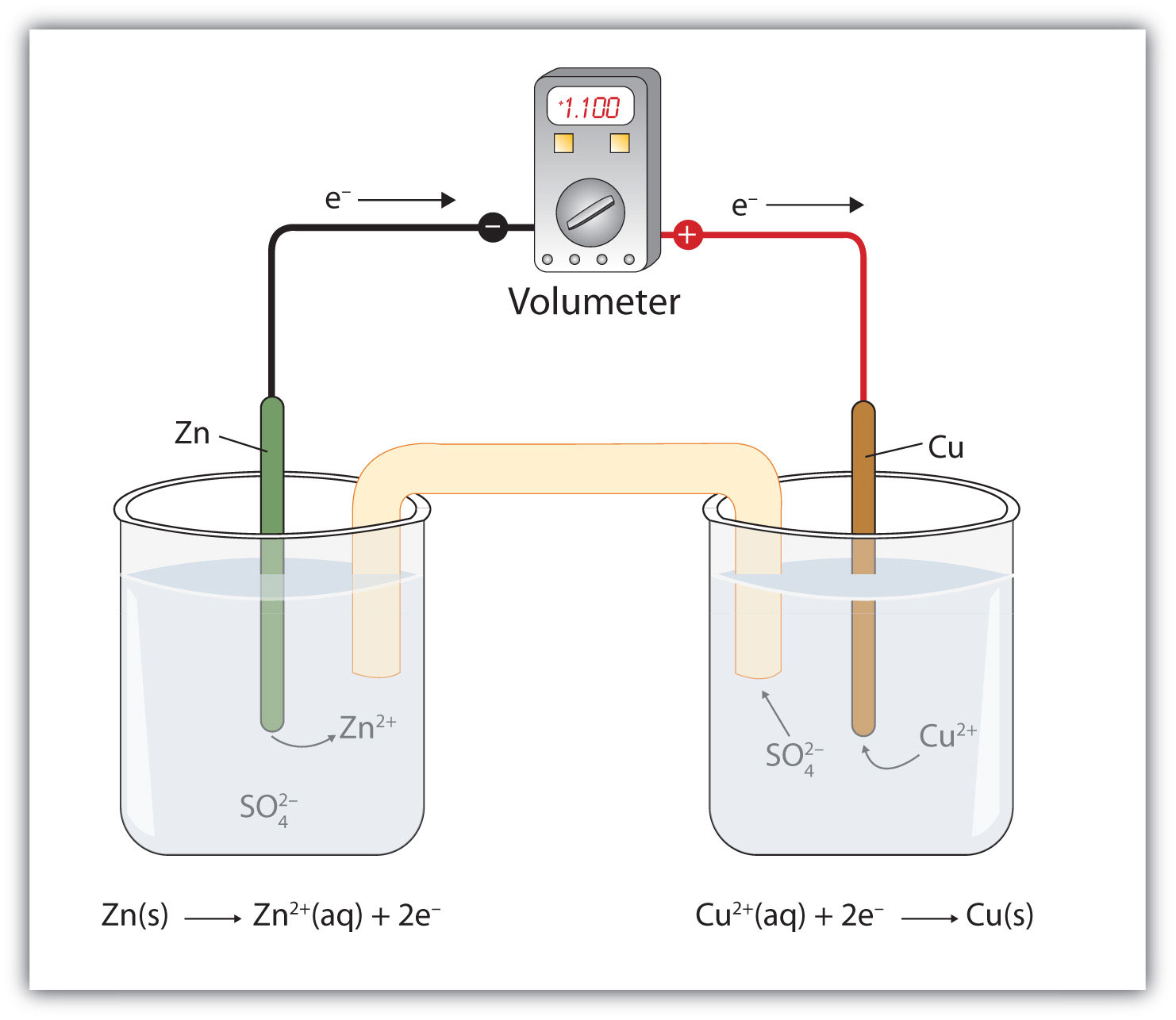
In order to accommodate such real-time changes, however, ESSs must be highly responsive to perturbations. This imbalance can be leveled out with ESSs. For example, the sporadic nature of renewable energy sources such as wind often results in oscillations in power supply, while certain times during the day require high levels of power. However, this is rarely the case in reality because fluctuations in both supply and demand occur. Ideally, power supply should match demand in order to maximize efficiency. ESSs can be used to stabilize the power grid through frequency regulation and peak shaving operations. 1 Introduction The recent push for renewable energy integration has shed light on the importance of grid-scale energy storage systems (ESSs). Finally, we offer some perspectives on the importance of optimizing zinc anodes as well as a future direction for developing high-performance aqueous zinc ion batteries. 
Furthermore, we discuss the energy density ramifications of the zinc anode with respect to its weight and reversibility through simple calculations for numerous influential reports in the field. This perspective offers a brief discussion of zinc electrochemistry in mildly acidic aqueous environments, along with an overview of recent efforts to improve the performance of zinc metal to extract key lessons for future research initiatives. In this regard, we aim to shed light on the importance of the zinc metal anode. However, many investigations often overlook the implications of the zinc metal anode, when in fact the anode is key to determining the energy density of the entire cell. This has led to aggressive research into viable intercalation cathodes, some of which have shown impressive results. As the anode is often fixed as zinc metal in these systems, most studies address the absence of a suitable cathode for reaction with zinc ions. The search has resulted in numerous candidates, among which mildly acidic aqueous zinc ion batteries have recently garnered significant academic interest, mostly due to their inherent safety. Despite the prevalence of lithium ion batteries in modern technology, the search for alternative electrochemical systems to complement the global battery portfolio is an ongoing effort. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
